AHCC® is produced by a patented process of Shiitake cell culture. AHCC®’s main active components, alpha-glucans, are small molecules easily absorbed by the organism. AHCC® is Japan’s top-selling immune support ingredient, its properties have been described in more than 60 scientific articles, including reports of more than 30 human clinical studies.
AHCC® is produced through a unique, patented fermentation process
AHCC® was developed in the late 1980’s by the Japanese biotechnology company Amino Up Co., in collaboration with scientists from the University of Tokyo. It started by the screening of hundreds of medicinal mushroom species, looking for those with the highest capacity to boost natural killer (NK) cell activity. A process of Shiitake cell culture was then developed, during which enzymes from the mushroom’s cells process the naturally occuring polysaccharides into small size alpha glucans.
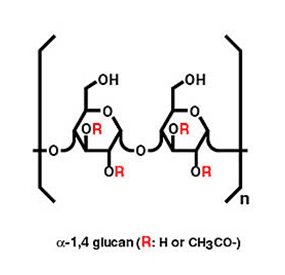
These unique, specific compounds confer remarkable properties to AHCC®, which has become Japan’s #1 specialty immune supplement and is used for a variety of applications including immune support, anti-viral defense, liver protection, reduction of cancer therapy side effects.
Regulation of immune system, protection against infections
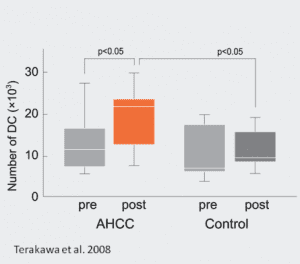
The potency of AHCC® as an immune system booster has been extensively documented. AHCC® stimulates the multiplication and activation of NK cells, which make one of the first line of defense of the immune system, and are major players in the fight against viral infections and cancer cells. It also increases the total number of dendritic cells, major regulators of cytokine production.
AHCC®’s immune boosting effects result in an increased protection against pathogens like Candida albicans, Pseudomonas, Staphylococcus aureus, as well as Influenza virus and West Nile virus.
A recent US study has also shown that AHCC® could be an efficient treatment against Human Papillomavirus infections.
AHCC and HPV – University of Texas study
Liver function and detoxification
Human clinical trials have shown that AHCC® can protect the liver against various types of damages: it improves the markers of liver damage in alcoholic fatty liver patients, and also had a protective effect in the context of hepatitis C infection. AHCC® protects liver cells against oxidative damage, inhibits the expression of pro-inflammatory gene iNOS. By supporting liver function, AHCC® allows a better elimination of toxins.
Intestinal health
AHCC® works as a prebiotic: it supports the growth of beneficial gut flora populations (Bifidobacteria). This is another, indirect mechanism by which AHCC® contributes to positively balance the immune system. AHCC® consumption improves gut epithelial cell function and results in a reduction of gut inflammation markers.
Safety of AHCC®
AHCC® has been used in more than 20 countries, for more than 20 years, and has been proven to be safe and devoid of side effects.
AHCC® Research Association
By clicking on the following links you will access the websites of the American and Japanese AHCC® research associations, which are working to coordinate and support the work of AHCC® researchers all over the world, and contribute to disseminate AHCC knowledge.
Japanese AHCC Research Association

Selected references
Aviles et al. 2008. Active hexose correlated compound activates immune function to decrease bacterial load in a murine model of intramuscular infection. Am J Surg. vol. 195(4), pp. 537-45.
Ito et al. 2014. Reduction of adverse effects by a mushroom product, active hexose correlated compound (AHCC) in patients with advanced cancer during chemotherapy – The significance of the levels of HHV-6 DNA in saliva as a surrogate biomarker during chemotherapy. Nut Cancer vol. 66(3), pp. 377-82.
Kim et al. 2014. Effect of active hexose correlated compound (AHCC) in alcohol-induced liver enzyme elevation. J Nutr Sci Vitaminol. vol. 60(5), pp. 348-56.
Matsui et al. 2011. Active hexose correlated compound inhibits the expression of proinflammatory biomarker iNOS in hepatocytes. Eur Surg Res. vol. 47(4), pp. 274-83.
Nogusa et al. 2009. Low dose supplementation with active hexose correlated compound improves the immune response to acute influenza infection in C57BL/6 mice. Nutr Res. vol. 29(2), pp. 139-43.
Roman et al. 2013. Short-term supplementation with active hexose correlated compound improves the antibody response to influenza B vaccine. Nutr Res. vol. 33(1), pp. 12-17.
Takanari et al. 2015. Effects of active hexose correlated compound on the seasonal variations of immune competence in healthy subjects. J Evid Based Complementary Altern Med. vol. 20(1), pp. 28-34.
Terakawa et al. 2008. Immunological effect of active hexose correlated compound (AHCC) in healthy volunteers: a double blind, placebo-controlled trial. Nutr Cancer vol. 60(5), pp. 643-51.
Yin et al. 2010. Effects of active hexose correlated compound on frequency of CD4+ and CD8+ T cells producing interferon-gamma and/or tumor necrosis factor-alpha in healthy adults. Hum Immunol. vol. 71(12), pp. 1187-90.
Graham et al. 2017. MicroRNA signature in the chemoprevention of functionally-enriched stem and progenitor pools (FESPP) by Active Hexose Correlated Compound (AHCC). Cancer Biol Ther. vol. 18(10), pp. 765-74.
Smith et al. 2019. From bench to bedside: evaluation of AHCC supplementation to modulate the host immunity to clear high-risk human papillomavirus infections. Front. Oncol. vol. 4(173).


